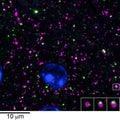
Anti-Gephyrin Antibody (L106/93)
Our Anti-Gephyrin mouse monoclonal primary antibody from NeuroMab is produced in-house from hybridoma clone L106/93. It detects human, mouse, and rat Gephyrin, and is purified by Protein A chromatography. It is great for use in AT, IHC, ICC, WB.
Human, Mouse, Rat
AT, ELISA, ICC, IHC, WB
Mouse
SKU: 75-444
Ships: 1-2 business days
Product Details
Gephyrin
In neuronal tissue, gephyrin is a scaffolding protein that self assembles in a complex, flat submembraneous lattice that inhibits mobility of the glycine receptors (GlyR) and GABAA receptors (GABAAR) causing clustering at post synaptic sites (Groeneweg et al, 2018). In non-neuronal tissue gephyrin plays a critical role in the molybdendum cofactor (MoCo) biosynthesis of essential life molybdoenzymes, like sulphite oxidase (Groeneweg et al, 2018). Three functional domains have been identified in gephyrin: the stable, structural G and E domains, and the C domain which is intrinsically unstructured leading to multiple isoforms (108, 105, 102, 98, 90 kDa) (Kawasaki, et al 1997). The 93 kDa protein predominantly expressed in the brain and located in the plasma membrane, has a 10X stronger affinity for the GlyR than the GABAAR. Gephyrin’s flexibility to change its size and molecular density is directly correlated to its high affinity to the GlyR-β subunit, and is required for anchoring and accurate clustering of GlyRs at post synaptic sites and microtubule transport chains (Greoneweg et al, 2018). A consistent parameter in the pathogenesis of Alzheimers Disease shows a decrease of inhibitory GABAergic synapses and gephyrin, and increased levels of an insoluble 37 kDa gephyrin fragment not detected in healthy, non-AD models (Kiss et al, 2016).
Purified by Protein A chromatography
1 mg/mL
Monoclonal
L106/93
IgG2a
AT, ELISA, ICC, IHC, WB
Mouse
GPHN GPH KIAA1385
80 kDa
Fusion protein amino acids 1-181 (N-terminus) of human Gephyrin (accession number Q9NQX3) produced recombinantly in E. Coli
Human
Human, Mouse, Rat
AB_2636852
Aliquot and store at ≤ -20°C for long term storage. For short term storage, store at 2-8°C. For maximum recovery of product, centrifuge the vial prior to removing the cap.
Liquid
Produced by in vitro bioreactor culture of hybridoma line followed by Protein A affinity chromatography. Purified mAbs are >90% specific antibody.
10 mM Tris, 50 mM Sodium Chloride, 0.065% Sodium Azide pH 7.125
Unconjugated
No cross-reactivity reported
Each new lot of antibody is quality control tested by western blot on rat whole brain lysate and confirmed to stain the expected molecular weight band.
These antibodies are to be used as research laboratory reagents and are not for use as diagnostic or therapeutic reagents in humans.
United States
24 months from date of receipt
Gephyrin [Includes: Molybdopterin adenylyltransferase (MPT adenylyltransferase) (EC 2.7.7.75) (Domain G); Molybdopterin molybdenumtransferase (MPT Mo-transferase) (EC 2.10.1.1) (Domain E)]
Shipped on ice packs
Product Specific References for Applications and Species
| Immunocytochemistry: Mouse | ||
| PMID | Dilution | Publication |
| 38684366 | 1:1000 | Matúš, D, et al. 2024. Essential Role of Latrophilin-1 Adhesion GPCR Nanoclusters in Inhibitory Synapses. The Journal of Neuroscience, . |
| 33083769 | 2ug/ml | Verstraelen, P., et al. 2020. Systematic Quantification of Synapses in Primary Neuronal Culture. iScience, 101542. |
| Western Blot: Mouse | ||
| PMID | Dilution | Publication |
| 33083769 | 1ug/ml | Verstraelen, P., et al. 2020. Systematic Quantification of Synapses in Primary Neuronal Culture. iScience, 101542. |