- Prepare samples and controls.
- Dilute 10X Injection Buffer 1:10 with 45 mL diH20.
- Reconstitute SR-FLIVO with 50 µL DMSO.
- Dilute SR-FLIVO 1:12 with 550 µL 1X Injection Buffer.
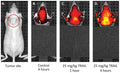
- Inject 100 µL intravenously.
- Let SR-FLIVO circulate 30-60 minutes.
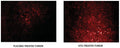
- View live tumor through a window chamber using a fluorescence microscope.
- If not viewing directly, excise tissue.
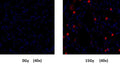
- If desired, label with additional stains, such as Hoechst 33342, or an antibody.
- If desired, fix cells.
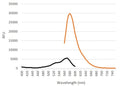
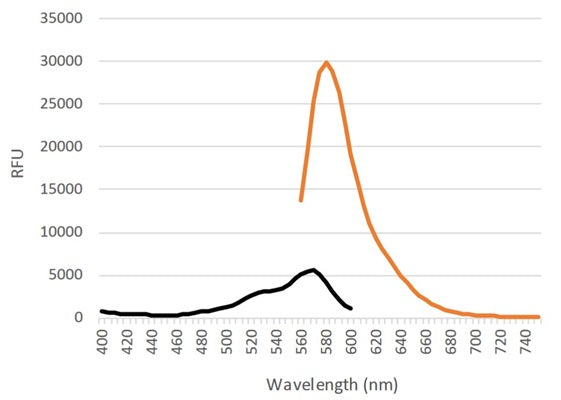
- Analyze with a fluorescence microscope, flow cytometer, or a window chamber system. SR-FLIVO excites at 550-580 nm and emits at 590-600 nm.
Thomas M, Davis T, Nell T, Sishi B, Engelbrecht A. Amino Acid Starvation Sensitizes Resistant Breast Cancer to Doxorubicin-Induced Cell Death. Front Cell Dev Biol. 2020 Oct 15;8:565915. doi: 10.3389/fcell.2020.565915. eCollection 2020. Full Text
"The body weight of the mice was monitored twice weekly. To assess intratumour caspase cleavage, FLIVOTM in vivo apoptosis tracers (Immunochemistry Technologies LLC, MN, United States) were used. The SR FLIVOTM red dye was prepared according to the manufacturer’s protocol and 100 μl was injected into the tail vein of mice after appropriate treatments were completed. After 1 h, whole tumors were excised, digested and analyzed using flow cytometry on the BD FACSAria I."
Kim JK, Byun MR, Maeng CH, Kim YR, Choi JW. Selective Targeting of Cancer Stem Cells (CSCs) Based on Photodynamic Therapy (PDT) Penetration Depth Inhibits Colon Polyp Formation in Mice. Cancers (Basel). 2020 Jan 14;12(1). pii: E203. doi: 10.3390/cancers12010203. Abstract
Kuchay, S;Giorgi, C;Simoneschi, D;Pagan, J;Missiroli, S;Saraf, A;Florens, L;Washburn, MP;Collazo-Lorduy, A;Castillo-Martin, M;Cordon-Cardo, C;Sebti, SM;Pinton, P;Pagano, M. PTEN counteracts FBXL2 to promote IP3R3- and Ca(2+)-mediated apoptosis limiting tumour growth. Nature. 2017 June 22. doi: 10.1038/nature22965. https://www.nature.com/nature/journal/vaop/ncurrent/full/nature22965.html. Full Article
Missiroli, S;Bonora, M;Patergnani, S;Poletti, F;Perrone, M;Gafà, R;Magri, E;Raimondi, A;Lanza, G;Tacchetti, C;Kroemer, G;Pandolfi, PP;Pinton, P;Giorgi, C. PML at Mitochondria-Associated Membranes Is Critical for the Repression of Autophagy and Cancer Development. Cell Report. 2016 August 30, Pages 2415-2427. doi:10.1016/j.celrep.2016.07.082. http://www.sciencedirect.com/science/article/pii/S2211124716310282. Full Article