Ships: 1-2 business days
- Prepare samples.
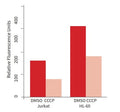
- Create controls with CCCP.
- Dilute 10X Assay Buffer 1:10 with diH2O.
- Reconstitute MitoPT® JC-1 with DMSO.
- Dilute MitoPT® JC-1 with 1X Assay Buffer.
- Add MitoPT® JC-1 to each sample.
- Incubate 15-30 minutes.
- Wash cells: add 1X Assay Buffer and spin cells (twice).
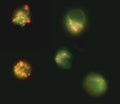
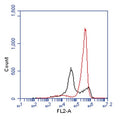
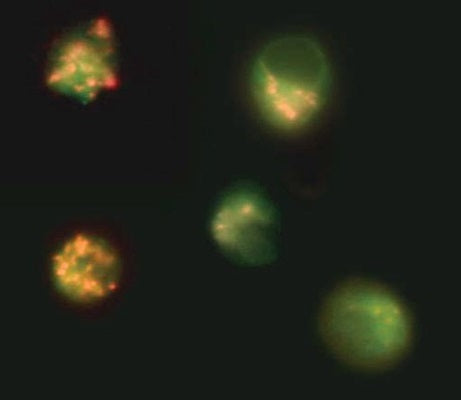
- Analyze with a fluorescence microscope, fluorescence plate reader, or flow cytometer. MitoPT® JC-1 excites at 488 nm. Aggregated MitoPT® JC-1 (orange) emits at 590 nm, monomeric MitoPT® JC-1 (green) emits at 527 nm.
Kit 911: 400 Tests
MitoPT® JC-1 Reagent, 400 Tests, #6260
10X Assay Buffer (2 x 125 mL), #6259
CCCP, 50 mM, 600 µL, #6258
Kit Manual
Kit 924: 100 Tests
MitoPT® JC-1 Reagent, 100 Tests, #6261
10X Assay Buffer, 60 mL, #685
CCCP, 50 mM, 125 µL, #6257
Kit Manual
Product Specific References
| PMID | Publication |
| 39845013 | Wang, Z, et al. 2024. Bullatine A suppresses glioma cell growth by targeting SIRT6. Heliyon, e41440-e41440. |
| 39251668 | Hajka, D., et al. 2024. Neuronal extracellular vesicles influence the expression, degradation and oligomeric state of fructose 1,6-bisphosphatase 2 in astrocytes affecting their glycolytic capacity. Scientific Reports, 20932. |
| 38414287 | Li, X, et al. 2024. Carnosol alleviates sepsis-induced pulmonary endothelial barrier dysfunction by targeting nuclear factor erythroid2-related factor 2/sirtuin-3 signaling pathway to attenuate oxidative damage. Phytotherapy research : PTR. |
| 38086312 | Magata, F., et al. 2023. Lipopolysaccharide impairs the in vitro growth, steroidogenesis, and maturation of oocyte-cumulus-granulosa cell complexes derived from bovine early antral follicles. Theriogenology, 187-194. |
| 38007509 | Cristaldi, M., et al. 2023. Caspase-8 activation by cigarette smoke induces pro-inflammatory cell death of human macrophages exposed to lipopolysaccharide. Cell death & disease, 773. |
| 37998320 | Tachibana, R., et al. 2023. Carboxylated Poly-L-lysine Potentially Reduces Human Sperm DNA Fragmentation after Freeze-Thawing, and Its Function Is Enhanced by Low-Dose Resveratrol. Cells. |
| 37504440 | Bingol Ozakpinar, O., et al. 2023. Carbon Nanofiber-Sodium Alginate Composite Aerogels Loaded with Vitamin D: The Cytotoxic and Apoptotic Effects on Colon Cancer Cells. Gels (Basel, Switzerland). |
| 35508969 | Kraemer, B.F., et al. 2022. Platelet mitochondrial membrane depolarization reflects disease severity in patients with preeclampsia. Molecular medicine (Cambridge, Mass.), 51. |
| 35965114 | Zhong, G., et al. 2022. Blood-brain barrier Permeable nanoparticles for Alzheimer's disease treatment by selective mitophagy of microglia. Biomaterials, 121690. |
| 35947192 | Yoshioka, H., et al. 2022. Deletion of Tfam in Prx1-Cre expressing limb mesenchyme results in spontaneous bone fractures. Journal of bone and mineral metabolism. |
| 36262666 | Funakoshi, A., et al. 2022. Cholecystokinin receptor antagonist suppresses melanoma growth by inducing apoptosis of tumor cells. JID Innovations, 100153. |
| 36331786 | Bülbül, B., et al. 2022. Novel 1,2,4-triazoles derived from Ibuprofen: synthesis and in vitro evaluation of their mPGES-1 inhibitory and antiproliferative activity. Molecular diversity. |
| 34193088 | Wu, W., et al. 2021. BCL-xL is Correlated With Disease Severity in Neonatal Infants With Sepsis. Research Square, 295. |