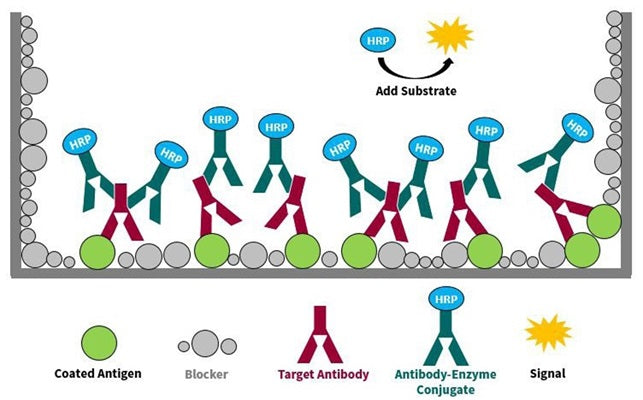
An Antigen-Down (AD) format ELISA refers to an ELISA plate configuration whereby the plate wells are pre-coated with a highly purified target antigen (hereafter called Target Antigen) that will subsequently be used to quantify specific antibody concentrations that are present within serological or cell culture origin samples (Figure 1). Having the ability to detect and quantitate a population of specific antibodies to a particular pathogen, allergen, or self-antigen within serological samples is essential for any successful epidemiological, allergic hypersensitivity, or autoimmune focused monitoring study.
Briefly, the specific antibody detection and quantification attributes of the AD ELISA format result from its simplistic design. Commercial ELISA plate manufacturers each utilize their own in-house polystyrene plastics modification process. Inner surfaces of each sample well possess the modified plastics surface chemistry to enable passive binding of the Target Antigen to the individual plate well surfaces. Examples of different types of Target Antigens include oligopeptides, polypeptides, and certain polysaccharide molecules. After the Target Antigen has been adsorbed to the ELISA plate wells, the entire well surface is treated with a blocking agent that will minimize unwanted non-specific binding signal. Reduction of non-specific binding signal is a universal goal of all types of ELISA format.
Once the Target Antigen-coated plate has been created (blocked and stabilized), screening of serum, plasma, or cell culture samples for the presence of a Target Antigen-specific poly or monoclonal antibody concentration or titer can begin. To do so, samples are incubated in the coated wells, after which the ELISA plates are washed to remove unbound sample. Next, anti-animal isotype specific IgG-HRP conjugate is added to each well and incubated. After the conjugate incubation period has ended, ELISA plate wells are washed to remove all non-specifically bound HRP conjugate. The next step is to add a chromogenic HRP substrate, such as TMB, to the plate wells. This will allow for the visualization and electronic quantitation of how much Target Antigen-specific antibodies were present within the samples. In the presence of the HRP-IgG conjugate (which is specifically bound to Target Antigen that was previously absorbed onto plate well surfaces), the horseradish peroxidase (HRP) will cause a color change in the initially clear TMB substrate solution. In general, the more intense the TMB color present within a respective sample well, the greater the amount of Target Antigen-specific antibody in that particular sample.
To summarize, the basic AD ELISA format is capable of providing a relative estimation of the Target Antigen-specific antibody titer/concentration present within the serological test samples, as well as a means by which to determine the identity of the isotype(s) that are participating in the humoral immune response. This Antigen- Down ELISA Development Kit was created to help guide end users through the common steps and frequently encountered issues associated with building an AD ELISA. The guide describes the assay performance milestones that must be achieved to enable progression through the various development stages of the antigen- down format ELISA (Table 1). The finalized ELISA should efficiently detect isotype-specific, low to high positive titer, humoral antibody responses in serum or plasma samples.