Ships: 1-2 business days
- Prepare samples and controls
- Dilute 10X Apoptosis Wash Buffer 1:10 with diH2O.
- Reconstitute FLICA with 50 µL DMSO.
- Dilute FLICA 1:5 by adding 200 µL PBS.
- Add diluted FLICA to each sample at 1:30-1:60 (e.g. spike at 1:30 by adding 10 µL to 290 µL cultured cells).
- Incubate approximately 1 hour.
- Wash and spin cells three times.
- If desired, label with additional stains, such as Hoechst, DAPI, or an antibody.
- If desired, fix cells.
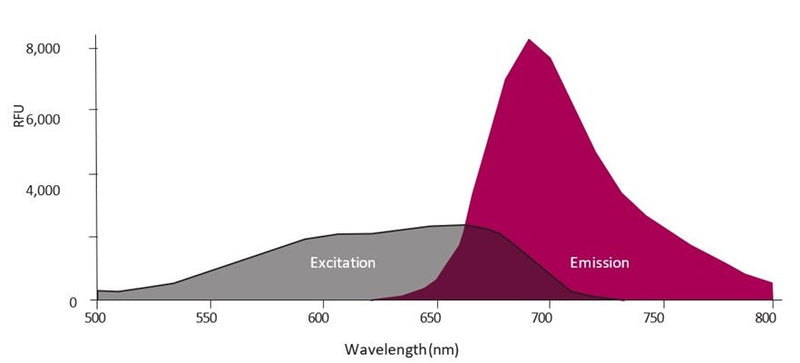
- Analyze with a fluorescence microscope or flow cytometer. FLICA 660 excites at 660 nm and emits at 680-690 nm.
Product Specific References
| PMID | Publication |
| 39843898 | Owens, SM, et al. 2025. Intrinsic p53 activation restricts gammaherpesvirus driven germinal center B cell expansion during latency establishment. Nature Communications, 951. |
| 37998361 | Spurlock, M., et al. 2023. The Inflammasome-Dependent Dysfunction and Death of Retinal Ganglion Cells after Repetitive Intraocular Pressure Spikes. Cells. |
| 35435672 | Li, Y, et al. 2022. Tumor Microenvironment-Responsive Yolk-Shell NaCl@Virus-Inspired Tetrasulfide-Organosilica for Ion-Interference Therapy via Osmolarity Surge and Oxidative Stress Amplification. ACS nano. |
| 35788117 | McLemore, AF, et al. 2022. Somatic gene mutations expose cytoplasmic DNA to co-opt the cGAS-STING-NLRP3 axis in Myelodysplastic syndromes. JCI insight. |
| 35967457 | Su, M, et al. 2022. Gasdermin D-dependent platelet pyroptosis exacerbates NET formation and inflammation in severe sepsis. Nature cardiovascular research, 732-747. |
| 36272266 | Deng, N, et al. 2022. Low-dose 5-fluorouracil ameliorates Th2 responses through the induction of apoptotic cell death of lung monocyte-derived dendritic cells in asthma. Biomedicine & pharmacotherapy, 113875. |
| 36373669 | Kodali, V, et al. 2022. Influence of Impurities from Manufacturing Process on the Toxicity Profile of Boron Nitride Nanotubes. Small (Weinheim an der Bergstrasse, Germany). |